NEBNext® Single Cell/Low Input RNA Library Prep Kit for Illumina®
Product information| Code | Name | Size | Quantity | Price | |
|---|---|---|---|---|---|
E6420S |
NEBNext Single Cell/Low Input RNA Library Prep Kit for Illumina |
24 rxns | - | Unavailable in your region | |
E6420L |
NEBNext Single Cell/Low Input RNA Library Prep Kit for Illumina |
96 rxns | - | Unavailable in your region |
NEBNext® Single Cell/Low Input RNA Library Prep Kit for Illumina®
Product Introduction
How low can you go?
NEBNext® is now available for single cell and ultra-low RNA inputs!
This unique workflow meets the demand for a highly sensitive, yet robust method that consistently enables generation of high quality sequencing data from single cell or ultra-low input RNA.
- Generate the highest yields of high-quality sequencing libraries from single cells, or as little as 2 pg - 200 ng RNA
- Experience unmatched detection of low abundance transcripts
- Rely on consistent transcript detection for a wide range of input amounts and sample types
- Observe uniform transcript coverage, regardless of input amount or sample type
- Use with a variety of RNA inputs, including cultured or primary cells, or total RNA
- Save time with a fast, streamlined workflow, minimal handling steps and hands-on time
| Catalog # | Size | Concentration |
|---|---|---|
| E6420S | 24 reactions | |
| E6420L | 96 reactions |
- Product Information
- Protocols, Manuals & Usage
- FAQs & Troubleshooting
- Citations & Technical Literature
- Quality, Safety & Legal
- Other Products You May Be Interested In
Product Information
Description
The NEBNext® Single Cell/Low Input RNA Library Prep Kit for Illumina® uses a template switching method to generate full length cDNAs directly from single cells or 2 pg – 200 ng RNA, followed by conversion to sequence-ready libraries using the Ultra™ II FS workflow. This unique workflow enables generation of the highest yields from a broad range of inputs, and superior transcript detection, while providing reliably consistent performance.
Please note that NEBNext Multiplex Oligos (Adaptors & Primers) are not included in the kit and are available separately.
Features:
- The highest yields of high-quality sequencing libraries from single cells, or 2 pg - 200 ng RNA
- Input can be cultured or primary cells, or total RNA
- Low abundance transcripts are easily detected
- High quality libraries and sequence data are generated for a wide range input amounts and sample types
- Full length transcript coverage
- Consistent transcript detection
- Superior transcript correlation
- Fast, streamlined, automation-friendly workflow, with minimal handling steps and hands-on time
- Single-tube protocol from cell lysis to cDNA
- DNA fragmentation, end repair and dA-tailing reagents in a single enzyme mix, with a single protocol, regardless of GC content
Download extensive performance data in our technical note.
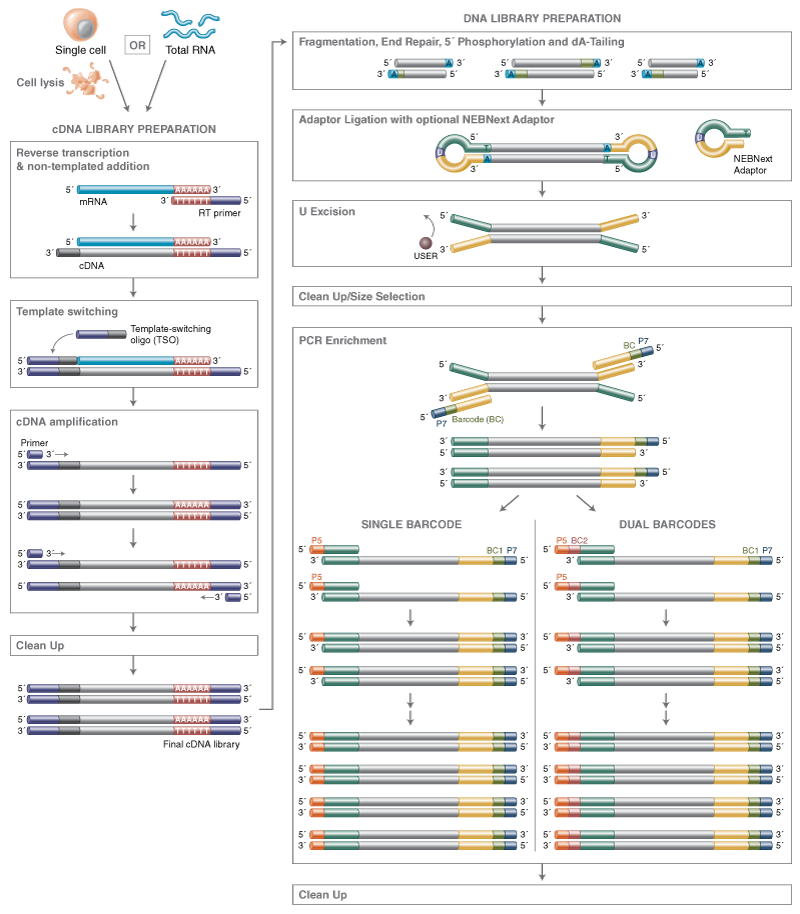
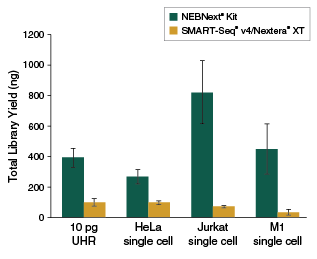
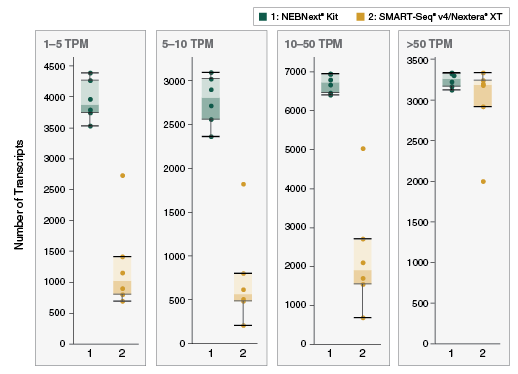
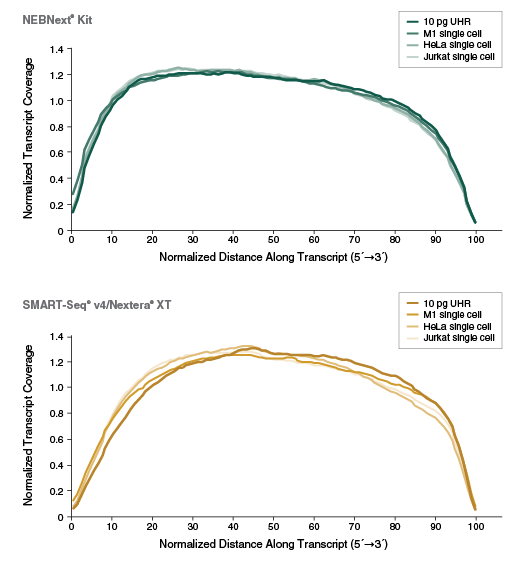
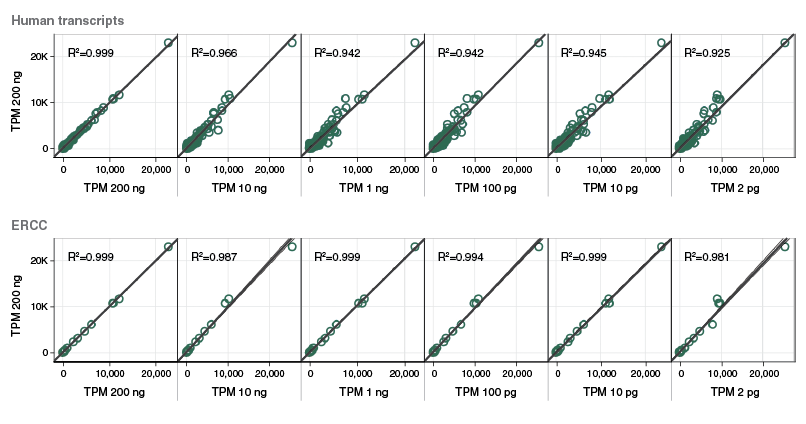
View additional performance data in our technical note.
- This product is related to the following categories:
- RNA Library Prep for Illumina,
- Next Generation Sequencing Library Preparation,
Kit Components
Kit Components
The following reagents are supplied with this product:
Properties & Usage
Materials Required but not Supplied
80% Ethanol (freshly prepared)Nuclease-free Water
DNA LoBind Tubes (Eppendorf® #022431021)
Magnetic rack or plate (e.g., NEBNext® Magnetic Separation Rack (NEB #S1515S), Alpaqua ® 96S Super Magnet Plate (#A001322), or equivalent)
Thermocycler
Vortex
Microcentrifuge
SPRIselect Reagent Kit (Beckman Coulter®, Inc. #B23317) or AMPure® XP
Beads (Beckman Coulter, Inc. #A63881)
Agilent® Bioanalyzer® or similar fragment analyzer and associated consumables
NEBNext Oligos
DNase RNase free PCR strip tubes (USA Scientific 1402-1708)
Protocols, Manuals & Usage
Manuals
FAQs & Troubleshooting
FAQs
- How much PBS can be carried over from cell collection into the cDNA Synthesis and Amplification protocol?
- What cell types does the NEBNext® Single Cell/Low Input RNA Library Prep Kit support?
- Can I resuspend the cells in buffers/medium other than PBS before cell collection?
- Do the cells have to be sorted in to a specific reagent?
- Can frozen cells be used?
- Can cells be lysed using other cell lysis buffers?
- How long can the synthesized cDNA (prior to cDNA amplification) be stored for prior to amplification?
- What types of RNA are compatible?
- Does the input total RNA have to be purified?
- My total RNA sample is in a volume > 8 µl. Can I still use the kit?
- Are the two PCR master mixes in the kit interchangeable?
- For low cDNA input do I have to do a double bead cleanup after the final PCR?
- How much cDNA input can I use for the NEBNext Ultra II FS DNA library preparation?
- What is the expected size range of libraries generated with the NEBNext Single Cell/Low Input RNA Library Prep Kit?
- Why are the adaptor dilution and library amplification PCR cycle number recommendations in this kit different from the NEBNext Ultra II FS DNA Library Prep Kit?
- Does the NEBNext Single Cell/Low Input RNA Library Prep Kit include adaptors and primers?
- How should the adaptor sequences be trimmed?
- Which NEBNext Oligos can be used with this library prep kit?
- Can I use this NEBNext kit with adaptors and primers from other vendors than NEB?
- Where can I find protocols for low-input RNA library prep for use with the following kits: NEBNext® Single Cell/Low Input RNA Library Prep Kit for Illumina (NEB #E6420), NEBNext® Single Cell/Low Input cDNA Synthesis & Amplification Module (NEB #E6421), and NEBNext® Single Cell Lysis Module (#E5530)?
Citations & Technical Literature
Citations
Additional Citations
- Mamanova L, et al. (2021) High-throughput full-length single-cell RNA-seq automation. Nat Protoc; DOI: 10.1038/s41596-021-00523-3
- Real E, et al. (2021) A single-cell atlas of Plasmodium falciparum transmission through the mosquito. Nat Commun; PubMedID: 34045457, DOI: 10.1038/s41467-021-23434-z
Quality, Safety & Legal
Quality Assurance Statement
Quality Control tests are performed on each new lot of NEB product to meet the specifications designated for it. Specifications and individual lot data from the tests that are performed for this particular product can be found and downloaded on the Product Specification Sheet, Certificate of Analysis, data card or product manual. Further information regarding NEB product quality can be found here.Specifications
The Specification sheet is a document that includes the storage temperature, shelf life and the specifications designated for the product. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]Certificate Of Analysis
The Certificate of Analysis (COA) is a signed document that includes the storage temperature, expiration date and quality controls for an individual lot. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]_[Lot Number]- E6420L_v1_10011961
- E6420S_v1_10011999
- E6420S_v1_10017871
- E6420S_v1_10023337
- E6420S_v1_10029225
- E6420L_v1_10030121
- E6420S_v1_10030119
- E6420S_v1_10033465
- E6420S_v1_10036493
- E6420L_v1_10037379
- E6420S_v1_10038641
- E6420S_v1_10042436
- E6420L_v1_10037360
- E6420S_v1_10033446
- E6420L_v1_10048315
- E6420S_v1_10049088
- E6420L_v1_10049123
- E6420L_v1_10052029
- E6420S_v1_10051750
- E6420S_v1_10059055
- E6420L_v1_10041376
- E6420L_v1_10061065
- E6420S_v1_10061938
- E6420S_v1_10062951
- E6420L_v1_10064654
- E6420S_v1_10065063
- E6420S_v1_10066990
- E6420L_v1_10056566
- E6420S_v1_10056554
- E6420L_v1_10078075
- E6420S_v1_10078077
- E6420L_v1_10082823
- E6420S_v1_10083180
- E6420L_v1_10091637
- E6420S_v1_10091660
- E6420L_v1_10093882
- E6420S_v1_10095647
- E6420S_v1_10085628
- E6420L_v1_10102745
- E6420L_v1_10085649
- E6420L_v1_10106630
- E6420S_v1_10106633
- E6420L_v1_10109862
- E6420S_v1_10113784
- E6420L_v1_10114862
- E6420S_v1_10099350
- E6420L_v1_10099364
- E6420L_v1_10126714
- E6420S_v1_10128255
- E6420L_v1_10131933
- E6420S_v1_10116848
- E6420L_v1_10135899
- E6420L_v1_10116866
- E6420S_v1_10151568
- E6420L_v1_10151645
- E6420L_v1_10155307
- E6420S_v1_10155605
- E6420S_v1_10159196
- E6420L_v1_10136369
- E6420S_v1_10165947
- E6420S_v1_10168330
- E6420L_v1_10168749
- E6420S_v1_10171970
- E6420L_v1_10174712
- E6420S_v1_10175370
- E6420L_v1_10177812
- E6420S_v1_10179080
- E6420L_v1_10159046
- E6420S_v1_10185791
- E6420L_v1_10186341
- E6420S_v1_10178947
- E6420L_v1_10206557
- E6420S_v1_10193476
- E6420L_v1_10193468
- E6420L_v1_10178927
- E6420S_v1_10213158
- E6420L_v1_10226172
- E6420L_v1_10226149
- E6420S_v1_10239655
- E6420L_v1_10231641
- E6420S_v1_10231654
- E6420L_v1_10260859
- E6420S_v1_10248002
- E6420L_v1_10275032
- E6420S_v1_10275643
- E6420G_v1_10265842
- E6420L_v1_10265852
- E6420S_v1_10265877
- E6420G_v1_10304320
- E6420L_v1_10290364
- E6420S_v1_10304322
- E6420S_v1_10290370
Safety DataSheets
The following is a list of Safety Data Sheet (SDS) that apply to this product to help you use it safely.NEBNext® Single Cell RT Primer Mix
NEBNext® Single Cell RT Buffer
NEBNext® Template Switching Oligo
NEBNext® Single Cell RT Enzyme Mix
NEBNext® Single Cell cDNA PCR Master Mix
NEBNext® Single Cell cDNA PCR Primer
NEBNext® Cell Lysis Buffer
Murine RNase Inhibitor
NEBNext® Bead Reconstitution Buffer
NEBNext® Adaptor Dilution Buffer
TE Buffer
Nuclease-free Water
NEBNext® Ligation Enhancer
NEBNext® Ultra™ II Ligation Master Mix
NEBNext® Ultra II Q5® Master Mix
NEBNext® Ultra™ II FS Enzyme Mix
NEBNext® Ultra™ II FS Reaction Buffer
Legal and Disclaimers
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
New England Biolabs (NEB) is committed to practicing ethical science – we believe it is our job as researchers to ask the important questions that when answered will help preserve our quality of life and the world that we live in. However, this research should always be done in safe and ethical manner. Learn more.
Licenses
This product is licensed for research and commercial use from Bio-Rad Laboratories, Inc., under U.S. Pat. Nos. 6,627,424, 7,541,170, 7,670,808, 7,666,645, and corresponding patents in other countries. No rights are granted for use of the product for Digital PCR or real-time PCR applications, with the exception of quantification in Next Generation Sequencing workflows.
For additional information or to inquire about commercial use, please contact busdev@neb.com.
Other Products You May Be Interested In
-

NEBNext® Multiplex Oligos for Illumina® (Dual Index Primers Set 2)
-

NEBNext® Multiplex Oligos for Illumina® (Index Primers Set 3)
-

NEBNext® Multiplex Oligos for Illumina® (96 Unique Dual Index Primer Pairs)
-

NEBNext® Multiplex Oligos for Illumina® (96 Index Primers)
-

NEBNext® Multiplex Oligos for Illumina® (Index Primers Set 1)
-

NEBNext® Multiplex Oligos for Illumina® (Dual Index Primers Set 1)
-

NEBNext® Multiplex Oligos for Illumina® (Index Primers Set 2)
-

NEBNext® Multiplex Oligos for Illumina® (Index Primers Set 4)
The supporting documents available for this product can be downloaded below.







