Luna® Universal Probe qPCR Master Mix
Product information| Code | Name | Size | Quantity | Price | |
|---|---|---|---|---|---|
M3004S |
Luna Universal Probe qPCR Master Mix |
200 rxns ( 2 X ) | - | Unavailable in your region | |
M3004L |
Luna Universal Probe qPCR Master Mix |
500 rxns ( 2 X ) | - | Unavailable in your region | |
M3004X |
Luna Universal Probe qPCR Master Mix |
1000 rxns (10x1ml) ( 2 X ) | - | Unavailable in your region | |
M3004E |
Luna Universal Probe qPCR Master Mix |
2500 rxns (1x25ml) ( 2 X ) | - | Unavailable in your region |
Luna® Universal Probe qPCR Master Mix
Product Introduction
Rapid, sensitive and precise probe-based qPCR detection and quantitation of target DNA and cDNA sequences.
Make a simpler choice
- One product per application simplifies selection
- Convenient master mix formats and user-friendly protocols simplify reaction setup
- Non-interfering, visible tracking dye helps to eliminate pipetting errors
Experience best-in-class performance
- All Luna® products have undergone rigorous testing to optimize specificity, sensitivity, accuracy and reproducibility
- Products perform consistently across a wide variety of sample sources
- A comprehensive evaluation of commercially-available qPCR and RT-qPCR reagents demonstrates superior performance of Luna products
- Read about the design of a multiplex assay to detect monkeypox virus DNA using Luna qPCR reagents
| Catalog # | Size | Concentration |
|---|---|---|
| M3004S | 200 reactions | 2 X |
| M3004L | 500 reactions | 2 X |
| M3004X | 1000 reactions | 2 X |
| M3004E | 2500 reactions | 2 X |
Featured Videos
View Video Library- Product Information
- Protocols, Manuals & Usage
- Tools & Resources
- FAQs & Troubleshooting
- Citations & Technical Literature
- Quality, Safety & Legal
- Other Products You May Be Interested In
Product Information
Description
Probe-based quantitative PCR (qPCR) uses real-time fluorescence released upon 5´→3´ exonuclease cleavage of a quenched, target-specific probe to measure DNA amplification at each cycle of a PCR. At a point where the fluorescence signal is significantly detectable over the background fluorescence, a quantification cycle or Cq value can be determined. Cq values can be used to evaluate relative target abundance between two or more samples or to calculate absolute target quantities in reference to an appropriate standard curve, derived from a series of known dilutions.
The Luna Universal Probe qPCR Master Mix is a 2X reaction mix optimized for real-time qPCR detection and quantitation of target DNA sequences using hydrolysis probes. It contains Hot Start Taq DNA Polymerase and has been formulated with a unique passive reference dye that is compatible across a variety of instrument platforms (including those that require a high or low ROX reference signal). It also features dUTP for carryover prevention and a non-fluorescent, visible dye to monitor reaction setup. This dye does not spectrally overlap fluorophores commonly used for qPCR and will not interfere with real-time detection.
The master mix formulation is supplied at 2X concentration and contains all PCR components required for amplification and quantitation of DNA except primers/probes and DNA template. Genomic DNA or cDNA of interest can be quantitated with Luna qPCR and existing as well as commercial qPCR assay primer/probe sequences can be used.
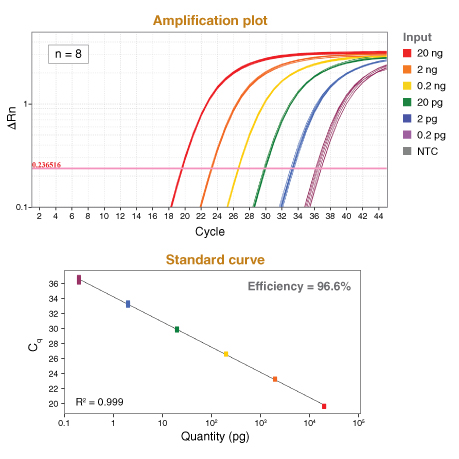
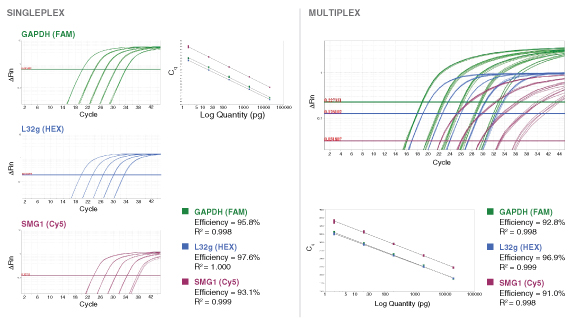
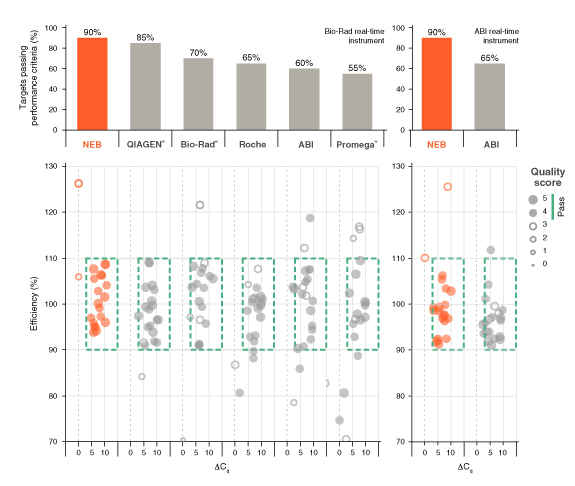
Learn more about our comprehensive qPCR/RT-qPCR testing and “dots in boxes” data visualization

- This product is related to the following categories:
- Luna® qPCR & RT-qPCR,
- PCR, qPCR & Amplification Technologies,
- This product can be used in the following applications:
- qPCR & RT-qPCR,
- DNA Amplification, PCR & qPCR
Reagents Supplied
Reagents Supplied
The following reagents are supplied with this product:
| NEB # | Component Name | Component # | Stored at (°C) | Amount | Concentration | |
|---|---|---|---|---|---|---|
Properties & Usage
Related Products
Companion Products
Product Notes
- Assay Design
The use of qPCR primer design software (e.g., Primer3) maximizes the likelihood of amplification success while minimizing nonspecific amplification and primer dimers. Targets with balanced GC/AT content (40–60%) tend to amplify efficiently. Where possible, enter sufficient sequence around the area of interest to permit robust primer design and use search criteria that permit cross-reference against relevant sequence databases (to avoid potential off-target amplification). For cDNA targets, it is advisable to design primers across known splicing sites in order to prevent amplification from genomic DNA. Conversely, primers designed to target intronic regions can ensure amplification exclusively from genomic DNA. - Primer and Probe Concentration
For most targets, a final concentration of 400 nM for each primer will provide optimum performance. If needed, primer concentrations can be optimized between 200–900 nM. Probe should be included at 200 nM for best results. Probe concentration can be optimized in the range of 100–500 nM if optimization of performance or target fluorescence level is desired. - Multiplexing
To detect or quantitate multiple targets in the same Luna reaction, select different fluorophores corresponding to separate detection channels of the real-time instrument. Include 400 nM of forward and reverse primer and 200 nM probe for each target to be detected in the reaction, and adjust concentrations if necessary based on performance (primer 200–900 nM, probe 100–500 nM). When loading qPCR protocol onto the real-time instrument, be sure to select the appropriate optical channels, as some instruments have a single channel recording mode that would prevent multiplex data collection and analysis. For ROX-dependent instruments, avoid ROX-labeled probes. The functionality of the primer and probe sets should be tested individually before attempting a multiplex reaction. When determining which fluorophores to include in a multiplex reaction, be sure to choose compatible reporter dyes and quenchers that have well separated fluorescence spectra or exhibit minimal overlap. - Amplicon Length
To ensure successful and consistent qPCR results, it is important to maximize PCR efficiency. An important aspect of this is the design of short PCR amplicons (typically 70–200 bp). Some optimization may be required (including the use of longer extension times), for targets that exceed that range. - Template Preparation and Concentration
Luna qPCR is compatible with DNA samples prepared through typical nucleic acid purification methods. Prepared DNA should be stored in an EDTA-containing buffer (e.g., 1X TE) for long-term stability, and dilutions should be freshly prepared for a qPCR experiment by dilution into either TE or water.
Generally, a useful concentration of standard and unknown material will be in the range of 106 copies to 1 copy. For gDNA samples from large genomes (e.g., human, mouse) a range of 50 ng–1 pg of gDNA is typical. For small genomes, adjust as necessary using 106 –1 copy input as an approximate range. Note that for single copy dilutions, some samples will contain multiple copies and some will have none, as defined by the Poisson distribution.
For cDNA, use the product of a reaction containing 1 μg–0.1 pg starting RNA. cDNA does not need to be purified before addition to the Luna reaction but should be diluted at least 1:10 into the qPCR. - ROX Reference Dye
Some real-time instruments recommend the use of a passive reference dye (typically ROX) to overcome well-to-well variations that could be caused by machine limitations such as “edge effect”, bubbles, small differences in volume, and autofluorescence from dust or particulates in the reaction. However, ROX normalization does little to the variations caused by pipetting errors of templates/primers, heterogeneous mixing, and evaporation/condensation issues.
A universal passive reference dye is included in the following Luna® qPCR products: Luna Universal qPCR Master Mix (NEB #M3003), Luna Universal Probe qPCR Master Mix (NEB #M3004), Luna Universal One-Step RT-qPCR Kit (NEB #E3005), and Luna Universal Probe One-Step RT-qPCR Kit (NEB #E3006). These products support broad instrument compatibility (High-ROX, Low-ROX, ROX-independent) so no additional ROX is required for normalization.
The Luna Probe One-Step RT-qPCR Kit (No ROX) (E3007) contains no reference dye and is compatible with any instrument that does not require ROX. If ROX normalization is needed, ROX can be added. Please refer to instrument manufacturer’s instructions for greater details.
- Carryover Contamination Prevention
qPCR is an extremely sensitive method, and contamination in new qPCR assays with products from previous amplification reactions can cause a variety of issues such as false positive results and a decrease in sensitivity. The best way to prevent this “carryover” contamination is to practice good laboratory procedures and avoid opening the reaction vessel post amplification. However, to accommodate situations where additional anti-contamination measures are desired, the Luna Universal Probe qPCR Master Mix contains a mixture of dUTP/dTTP that results in the incorporation of dU into the DNA product during amplification. Pretreatment of qPCR experiments with uracil DNA glycosylase (UDG) will eliminate previously-amplified uracil-containing products by excising the uracil base to produce a non-amplifiable DNA product. The use of a thermolabile UDG is important, as complete inactivation of the UDG is required to prevent destruction of newly synthesized qPCR products.
To enable carryover prevention, 0.025 units/μl Antarctic Thermolabile UDG (NEB #M0372) should be added to the reaction mix. To maximize elimination of contaminating products, set up the qPCR experiments at room temperature or include a 10 minute incubation step at 25°C before the initial denaturation step. - Reaction Setup and Cycling Conditions
Due to the hot start nature of the polymerase, it is not necessary to preheat the thermocycler prior to use or set up reactions on ice.
For 96-well plates, we recommend a final reaction volume of 20 μl.
For 384-well plates, a final reaction volume of 10 μl is recommended.
When programming instrument cycling conditions, ensure a plate read is included at the end of the extension step, and a denaturation (melt) curve after cycling is complete to analyze product specificity.
Amplification for 40 cycles is sufficient for most applications, but for very low input samples 45 cycles may be used.
Protocols, Manuals & Usage
Protocols
Manuals
Usage & Guidelines
Application Notes
Tools & Resources
Web Tools
FAQs & Troubleshooting
FAQs
- How do I use qPCR to determine the concentration of my material?
- Can I set up my Luna® qPCR at room temperature?
- What is the difference between probe- and dye-based versions of the Luna® qPCR Mixes?
- Should I use probe- or dye-based detection for my qPCR assays?
- How should I design primers for Luna® qPCR?
- How long should my amplicon be for qPCR?
- Why is the Luna® qPCR Mix blue? Will this dye interfere with detection?
- Can I run the Luna® qPCR Mix on my qPCR instrument?
- Can I use fast instrument settings with the Luna® qPCR Mix?
- Do I need to add ROX?
- How many dilutions should I use to make a standard curve?
- Why does NEB recommend 40-45 cycles?
- Does the Luna® qPCR Mix contain dUTP? Can I use carryover contamination prevention methods?
- Can I run multiplex reactions with the Luna® Universal Probe Master Mix? Do I need to change my reaction conditions?
- Can I use a ROX-labeled probe with the Luna® Probe Mixes that contain a universal ROX reference dye?
- Can alternative probe based detection strategies be used with the Luna® Probe Mix?
- What samples can be used in qPCR with the Luna® Mix?
- Can I use cDNA? Does it matter how I make it?
- How much template material can I use in Luna® qPCR?
- How much primer and probe should I use with the Luna® Universal Probe qPCR Master Mix?
- Can I use shorter cycling times?
Troubleshooting
Citations & Technical Literature
Citations
Additional Citations
Quality, Safety & Legal
Quality Assurance Statement
Quality Control tests are performed on each new lot of NEB product to meet the specifications designated for it. Specifications and individual lot data from the tests that are performed for this particular product can be found and downloaded on the Product Specification Sheet, Certificate of Analysis, data card or product manual. Further information regarding NEB product quality can be found here.Specification Change Notifications
Specifications
The Specification sheet is a document that includes the storage temperature, shelf life and the specifications designated for the product. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]Certificate Of Analysis
The Certificate of Analysis (COA) is a signed document that includes the storage temperature, expiration date and quality controls for an individual lot. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]_[Lot Number]- M3004S_L_G_v1_0021611
- M3004S_L_G_v1_0031701
- M3004S_L_G_X_E_v1_0041706
- M3004S_L_G_X_E_v1_0051709
- M3004S_L_G_X_E_v1_0061801
- M3004S_v1_10011154
- M3004E_v1_10012649
- M3004L_v1_10012025
- M3004S_v1_10012578
- M3004X_v1_10011846
- M3004L_v1_10014397
- M3004S_v1_10014514
- M3004L_v1_10015718
- M3004X_v1_10015719
- M3004L_v1_10017708
- M3004S_v1_10017705
- M3004X_v1_10017680
- M3004G_v1_10020125
- M3004E_v1_10026311
- M3004L_v1_10021860
- M3004X_v1_10025599
- M3004X_v1_10028158
- M3004S_v1_10029048
- M3004X_v1_10029066
- M3004X_v1_10029382
- M3004E_v1_10031120
- M3004S_v1_10030035
- M3004X_v1_10029509
- M3004S_v1_10031521
- M3004S_v1_10031924
- M3004X_v1_10031773
- M3004X_v1_10032190
- M3004L_v1_10029423
- M3004X_v1_10033570
- M3004X_v1_10034023
- M3004L_v1_10034021
- M3004X_v1_10034233
- M3004X_v1_10034480
- M3004L_v1_10036689
- M3004S_v1_10036692
- M3004X_v1_10036698
- M3004G_v1_10020285
- M3004G_v1_10036063
- M3004S_v1_10038444
- M3004G_v1_10036694
- M3004S_v1_10039129
- M3004X_v1_10038768
- M3004X_v1_10041840
- M3004L_v1_10038445
- M3004X_v1_10044566
- M3004X_v1_10045782
- M3004E_v1_10045787
- M3004L_v1_10045783
- M3004S_v1_10043814
- M3004X_v1_10048006
- M3004E_v1_10048832
- M3004L_v1_10048007
- M3004X_v1_10048534
- M3004S_v1_10051186
- M3004E_v1_10051853
- M3004X_v1_10051224
- M3004E_v1_10053483
- M3004S_v1_10054119
- M3004X_v1_10053283
- M3004G_v1_10055844
- M3004E_v1_10055967
- M3004L_v1_10053282
- M3004X_v1_10056172
- M3004E_v1_10058000
- M3004G_v1_10058597
- M3004X_v1_10057599
- M3004G_v1_10059566
- M3004L_v1_10057742
- M3004S_v1_10057923
- M3004X_v1_10059071
- M3004X_v1_10061400
- M3004E_v1_10061494
- M3004L_v1_10061473
- M3004X_v1_10062495
- M3004L_v2_10064605
- M3004S_v1_10062519
- M3004X_v1_10064599
- M3004E_v1_10064056
- M3004L_v2_10067377
- M3004E_v2_10071098
- M3004L_v2_10071659
- M3004S_v2_10067379
- M3004X_v2_10069945
- M3004L_v2_10072588
- M3004S_v2_10071660
- M3004X_v2_10072587
- M3004E_v2_10076308
- M3004E_v2_10077666
- M3004S_v2_10078045
- M3004L_v2_10079204
- M3004S_v2_10081643
- M3004E_v2_10081644
- M3004E_v2_10085117
- M3004S_v2_10084016
- M3004L_v2_10083897
- M3004E_v2_10087140
- M3004S_v2_10088140
- M3004G_v2_10067092
- M3004L_v2_10088139
- M3004X_v2_10088141
- M3004L_v2_10090738
- M3004S_v2_10089098
- M3004X_v2_10090728
- M3004E_v2_10090578
- M3004S_v2_10091415
- M3004L_v2_10094024
- M3004S_v2_10094951
- M3004E_v2_10094674
- M3004X_v2_10094088
- M3004L_v2_10097876
- M3004S_v2_10097899
- M3004X_v2_10097878
- M3004E_v2_10097879
- M3004S_v2_10100714
- M3004L_v2_10100722
- M3004X_v2_10100716
- M3004L_v2_10104272
- M3004S_v2_10104389
- M3004X_v2_10104271
- M3004S_v2_10105026
- M3004E_v2_10104400
- M3004X_v2_10107017
- M3004G_v2_10104393
- M3004S_v2_10108354
- M3004S_v2_10109266
- M3004S_v2_10114330
- M3004S_v2_10116093
- M3004L_v2_10119623
- M3004X_v2_10122287
- M3004L_v2_10125226
- M3004X_v2_10125225
- M3004S_v2_10116349
- M3004S_v2_10128182
- M3004X_v2_10129770
- M3004S_v2_10131640
- M3004L_v2_10129771
- M3004X_v2_10131891
- M3004S_v2_10133730
- M3004E_v1_10134388
- M3004E_v1_10113576
- M3004E_v1_10119832
- M3004G_v1_10125473
- M3004G_v1_10127759
- M3004G_v2_10109265
- M3004L_v2_10107693
- M3004L_v2_10113699
- M3004L_v2_10135832
- M3004S_v2_10137988
- M3004X_v2_10107692
- M3004X_v2_10113700
- M3004X_v2_10135833
- M3004S_v2_10138852
- M3004S_v2_10140111
- M3004L_v2_10140465
- M3004X_v2_10140464
- M3004E_v1_10135830
- M3004G_v2_10142512
- M3004E_v1_10143237
- M3004E_v1_10145285
- M3004L_v2_10145858
- M3004X_v2_10145859
- M3004S_v2_10146145
- M3004L_v2_10148915
- M3004L_v2_10150281
- M3004X_v2_10150282
- M3004E_v1_10153118
- M3004X_v2_10153104
- M3004L_v2_10154275
- M3004X_v2_10155828
- M3004G_v2_10157110
- M3004G_v1_10158319
- M3004E_v1_10161035
- M3004S_v2_10154277
- M3004L_v2_10157231
- M3004X_v2_10159708
- M3004G_v2_10161569
- M3004E_v1_10164558
- M3004X_v2_10163243
- M3004E_v1_10166600
- M3004L_v2_10164982
- M3004X_v2_10166799
- M3004E_v1_10171019
- M3004L_v2_10170225
- M3004X_v2_10170224
- M3004S_v2_10164984
- M3004E_v1_10176716
- M3004L_v2_10175345
- M3004X_v2_10175344
- M3004E_v1_10180140
- M3004L_v2_10182992
- M3004X_v2_10182991
- M3004E_v1_10186902
- M3004L_v2_10189288
- M3004S_v2_10195523
- M3004L_v2_10200220
- M3004X_v2_10200223
- M3004E_v1_10200218
- M3004S_v2_10200221
- M3004L_v2_10203475
- M3004X_v2_10203474
- M3004E_v1_10204372
- M3004L_v2_10206927
- M3004X_v2_10206928
- M3004G_v1_10200219
- M3004S_v2_10206926
- M3004E_v1_10210693
- M3004G_v1_10214151
- M3004L_v2_10210964
- M3004X_v2_10211738
- M3004S_v2_10219575
- M3004E_v1_10221852
- M3004L_v2_10220975
- M3004X_v2_10220974
- M3004G_v2_10221843
- M3004L_v2_10230397
- M3004S_v2_10230400
- M3004G_v1_10234516
- M3004E_v1_10233909
- M3004X_v2_10234112
- M3004E_v1_10238397
- M3004S_v2_10238408
- M3004G_v1_10236415
- M3004G_v1_10242548
- M3004X_v2_10244387
- M3004S_v2_10246822
- M3004E_v1_10247110
- M3004X_v2_10250248
- M3004S_v2_10255846
- M3004E_v1_10255845
- M3004L_v2_10255844
- M3004X_v2_10258437
- M3004S_v2_10262326
- M3004X_v2_10262324
- M3004L_v2_10267529
- M3004X_v2_10272650
- M3004S_v2_10272649
- M3004G_v2_10280333
- M3004E_v1_10276408
- M3004L_v2_10279050
- M3004E_v1_10284181
- M3004X_v2_10284180
- M3004L_v2_10284184
- M3004S_v2_10282748
- M3004G_v2_10287823
- M3004E_v1_10291487
- M3004G_v2_10294173
- M3004S_v2_10291315
- M3004X_v2_10293024
- M3004L_v2_10300263
- M3004E_v1_10295771
- M3004X_v2_10304231
Safety DataSheets
The following is a list of Safety Data Sheet (SDS) that apply to this product to help you use it safely.Luna® Universal Probe qPCR Master Mix
Legal and Disclaimers
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
New England Biolabs (NEB) is committed to practicing ethical science – we believe it is our job as researchers to ask the important questions that when answered will help preserve our quality of life and the world that we live in. However, this research should always be done in safe and ethical manner. Learn more.
Licenses
Nucleic acid-based aptamers for use with thermophilic DNA polymerases are licensed exclusively by New England Biolabs, Inc. from SomaLogic, Inc. New England Biolabs, Inc. gives the Buyer/User a non-exclusive license to use the Luna Universal Probe qPCR Master Mix for Research Use Only (RUO). Commercial use of this product may require a license from New England Biolabs, Inc. For additional information or to inquire about commercial use, please contact busdev@neb.com.Trademarks
BIO-RAD® is a registered trademark of Bio-Rad Laboratories.FASTSTART™ is a trademark of Roche.
GOTAQ® and PROMEGA® are registered trademarks of Promega Corporation.
QIAGEN® and QUANTITECT® are registered trademarks of Qiagen, Inc.
QUANTSTUDIO® is a registered trademark of Life Technologies Corporation.
SOMAMER® is a registered trademark of Somalogic, Inc.
SSOADVANCED™ is a trademark of Bio-Rad Laboratories.
STEPONE® is a registered trademark of Applied Biosystems, LLC.
SYBR® is a registered trademark of Molecular Probes, Inc.
TAQMAN® is a registered trademark of Thermo Fisher Scientific.
Other Products You May Be Interested In
The supporting documents available for this product can be downloaded below.





